Difference Between Reversible Process and Irreversible Process in Thermodynamics [PDF]

Hello readers, in this article, we will be going to see the Difference between Reversible Process and Irreversible Process in Thermodynamics and I hope you can clear this concept by reading this paper.
So before jumping into the differences, let me give you the general idea of What is Reversible and Irreversible processes in thermodynamics.
Reversible Process:
Reversible Process is a process that can be made to exactly replace its path without suffering any deviation. A process in which there are no losses is called Reversible Process.
For Example,
- Spring-mass system
- Electroplating
So let me give you an example of this with a simple equation! Suppose:
A + B = C + D
And if the Reverse is attained that is C + D= A + B then that process is called Reversible Process.
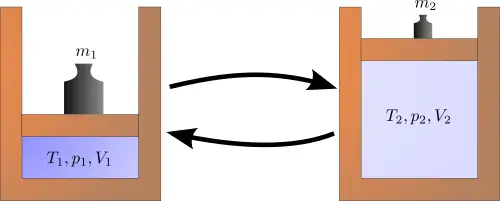
Irreversible process:
An irreversible process is a process that cannot be made to exactly retrace its path without suffering its deviation is called an irreversible process.
Most of the processes in nature are irreversible. Any process with friction or losses is called an irreversible process.
For example,
- Sun’s energy coming to earth.
- A hot cup of coffee kept on the table
- combustion
- Wind flow.
So Now Let's Dive into the Differences of Reversible Process and Irreversible Process:
| Reversible Process | Irreversible Process |
|---|---|
| Reversible Process is a process that can be made to exactly replace its path without suffering any deviation. | An irreversible process is a process that cannot be made to exactly retrace its path without suffering its deviation |
| A process in which there are no losses is called Reversible Process. | A process with friction or losses is called an irreversible process. |
| Reversible process can be carried out very slowly. | The irreversible process can be carried out fastly. |
| Maximum work can be obtained in this process. | Maximum work cannot be obtained in this process. |
| This process can be reversed called Reversible Process. | This process cannot be reversed called Irreversible Process. |
| This process can take infinite time for completion. | This process can take a finite time for completion. |
| Examples of Reversible Process are Spring-mass system, Electroplating, etc. | Examples of Irreversible Processes are A hot cup of coffee kept on the table, Combustion process, and wind flow. |
This is the complete explanation about the Difference between Reversible Process and irreversible process which is shown in a detailed manner. If you have any doubts, feel free to ask us in the comments section.
More Resources:
What is the Difference Between Diathermic and Adiabatic Process?
Types of Thermodynamic System with Example
Difference Between Nano, Macro, Micro and Smart Material